Effect of TU-100, a traditional Japanese medicine, administered after hepatic resection in patients with liver cancer: a multi-center, phase III trial (JFMC40-1001) - International Journal of Clinical Oncology
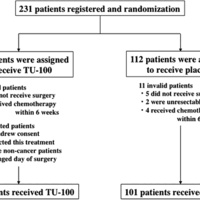
Background This multi-center, phase III trial assesses the efficacy of daikenchuto (TU-100) on gastrointestinal disorders after hepatic resection (UMIN Registration No. 000003103). Materials and methods A total of 231 patients, who underwent hepatic resection at 26 Japanese centers, were enrolled. …